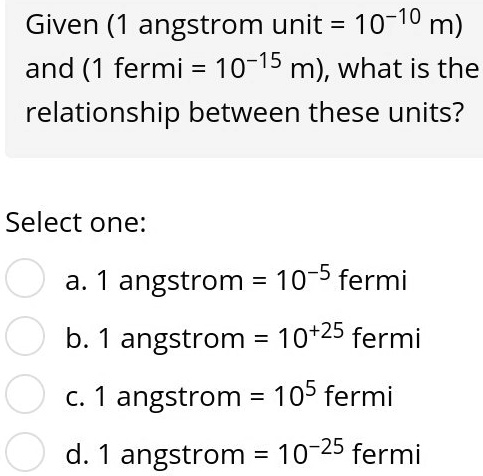
SOLVED: What is thc - wavelength (angstroms) of a photon that has an energy of 5.69 * 10-17 J? Angstrom Ixlo-10 m) 454 angstroms 6,89 10L5 angstroms 34.9 angstroms 3.66 109 angstroms 3.50 10-9 angstroins

How would you measure…. A Football field? A drive in the car? Your weight? Kool-aid? UNITS! - ppt download

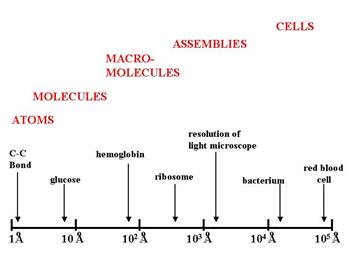
1Åm = 1×10 −10 m. The ångström is often used in natural sciences and technology to express the sizes of atoms, molecules, and microscopic biological structures, - ppt download
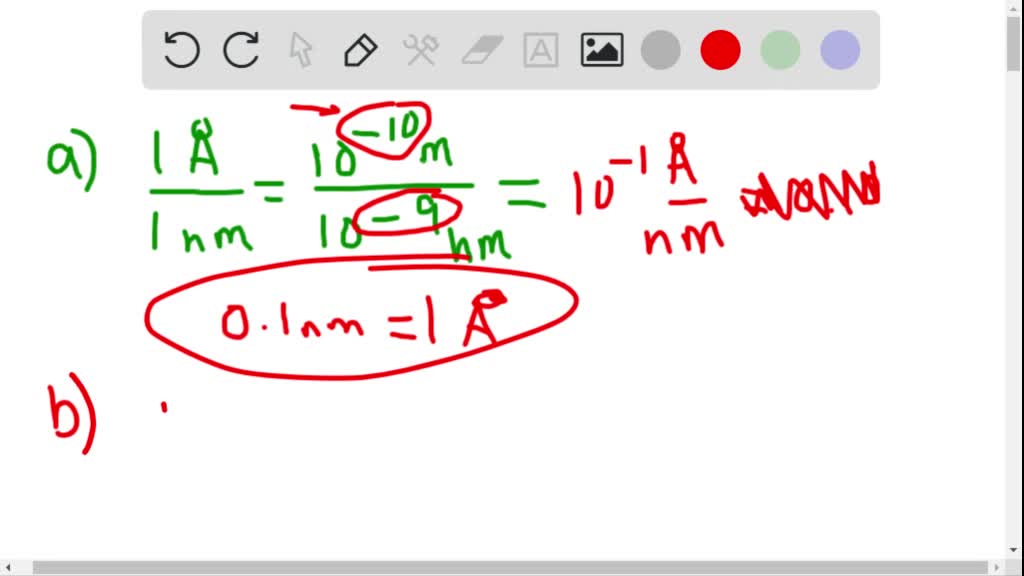
SOLVED:An angstrom (symbol  ) is a unit of length, defined as 10^-10 m, which is on the order of the diameter of an atom. (a) How many nanometers are in 1.0
The Bell System technical journal . th lattice constant a Angstroms, Oo = a^l2. Substituting the expressionfor N{E) into the equation for Ep gives Ef = Sraax. = 21.6fio-2/3ev. (v) Expressing


![Convert Meter [m] to Angstrom [Å] - Distance Conversion Convert Meter [m] to Angstrom [Å] - Distance Conversion](https://www.dailytoolz.com/convert-meter-to-angstrom/img/pageimage.jpg)


:max_bytes(150000):strip_icc()/hand-touching-two-points-on-digital-ruler-164210744-58a39ea43df78c475847a2f5.jpg)